4e édition de la Journée Scientifique Lumomat
L’École Universitaire de Recherche Lumomat a la plaisir de vous inviter à sa 4ème Journée Scientifique qui se déroulera le Mercredi 20 Mai 2026 à Nantes.
📆 Programme de la journée
- 4 conférences plénières animées par des chercheurs et chercheuses internationaux.
- Des communications orales des doctorantes et doctorants du réseau Lumomat (sur sélection).
- Une session posters (sur simple inscription).
➖ Conférences internationales
Erick L. Bastos (Institute of Chemistry - University of São Paulo (IQ-USP)) - Brésil

Nature’s Colorful Palette: Functional Betalain Photochemistry
This talk explores the biofluorescence of plant pigments, with emphasis on betalains as versatile scaffolds for functional molecular systems and advanced materials. We investigate the photophysical and photochemical behavior of these natural products and their pseudo-natural derivatives to establish structure–property relationships. These insights have enabled the development of safe colorants and fluorescent dyes, tunable superoxide generators, and self-assembling chiral systems that direct energy transfer. Together, these advances illustrate sustainable strategies for sensing, imaging, redox chemistry, and energy conversion.
Inés Corral Pérez (Autonomous Univeristy of Madrid) - Espagne

Light-Driven Chemistry from Molecular Origins to Functional Nanomaterials Organic triplet photosensitizers: molecular design, mechanisms and applications
Electronic excited states define how molecules survive, transform, and function upon light absorption. From the photostability of DNA to the optical response of functional nanomaterials, understanding these excited-state processes is essential to rationalizing both biological resilience and optoelectronic performance.
In DNA, the remarkable photostability of canonical nucleobases is rooted in the topography of their excited-state potential energy surfaces. Ultrafast internal conversion pathways efficiently funnel the excited population back to the ground state, minimizing excited-state lifetimes, fluorescence, and triplet formation. By systematically exploring substitution effects in purine and pyrimidine derivatives, we identify the key structural and electronic factors that differentiate canonical nucleobases from alternative heterocycles and underpin their photophysical robustness. [1–3]
At the other end of the molecular complexity spectrum, π-conjugated nanographenes exhibit tunable band gaps, rich emission profiles, and promising optoelectronic functionalities. Curvature, helicene constraints, and supramolecular organization introduce multiple deactivation channels, including thermally activated delayed fluorescence and efficient singlet oxygen generation. These structural features give rise to intricate excited-state landscapes that directly govern their optical properties. [4,5]
Using advanced quantum chemical approaches and excited-state simulations, we uncover the relaxation pathways that govern light-induced processes in both nucleobases and nanographenes. By revealing the excited-state principles underlying molecular photostability and functional photophysics, these studies provide a coherent framework to understand how structure shapes light-induced dynamics across chemically diverse π-systems.
References: [1] C. E. Crespo-Hernández, L. Martínez-Fernández, C. Rauer, C. Reichardt, S. Mai, M. Pollum, P. Marquetand, L. González, I. Corral, J. Am. Chem. Soc. 2015, 137, 4368. [2] E. Vos, S. J. Hoehn, S. E. Krul, C. E. Crespo-Hernández, J. González-Vázquez, I. Corral, J. Phys. Chem. Lett. 13, 2022, 2000. [3] L. Martínez-Fernández, I. Corral, Excited States Reactivity of Natural and Unnatural DNA Nucleobases in Comprehensive Computational Chemistry, Volume 4, 2024, 306-329, Elsevier [4] S. Ramírez-Barroso, F. Romeo-Gella, J. M. Fernández-García, S. Feng, L. Martínez-Fernández, D. García-Fresnadillo, I. Corral, N. Martín, R. Wannemacher, Adv. Mat. 2023, 35, 2212064.
Mariangela Di Donato (The Institute of Chemistry of OrganoMetallic Compounds / LENS-European Laboratory for Nonlinear Spectroscopy) - Italie

Organic triplet photosensitizers: molecular design, mechanisms and applications
The design and synthesis of organic compounds presenting efficient Inter System Crossing (ISC) and long-lived triplet excited states holds significant importance for multiple applications, encompassing photovoltaics, photocatalysis, photodynamic therapy and biosensing. Nevertheless, our understanding of the photophysical mechanisms underpinning the formation and temporal evolution of triplets remains incomplete. Efficient ISC usually requires the use of heavy atoms, posing problems about toxicity and increased synthetic costs, while the realization of fully organic supramolecular structures efficiently producing long lived triplet states upon the absorption of visible light still represents a challenging goal. We characterized different triplet photosensitizers (PSs) devoid of heavy atoms, whose molecular structure is meticulously optimized to amplify spin-orbit coupling, thereby enhancing ISC efficiency through various promising mechanisms, including Charge Transfer-ISC [1], Symmetry Breaking-ISC [2], and specific molecular design strategies tailored to stabilize the triplet state [3]. We employ a comprehensive approach to investigate the photochemical properties of these compounds, involving various steady state and time-resolved spectroscopy techniques, cyclic voltammetry, and density functional theory (DFT) calculations. Our studies provide significant advances toward the realization of highly efficient triplet PSs, whose electronic properties can be fine-tuned to align with the requirements of specific applications.
References : [1] J. Deckers et al, J. Mater. Chem. C, 2022, 10, 9344; Z Wang, et al, Chemistry–A European Journal, 2020, 26, 1091-1102. [2] Y Liu et al, The Journal of Physical Chemistry C, 2018, 122, 2502-2511 [3] D. Liu et al, Angew. Chem. Int. Ed. 2020, 59, 11591 – 11599
Timothy Barendt (School of Chemistry - University of Birmingham) - Royaume-Uni

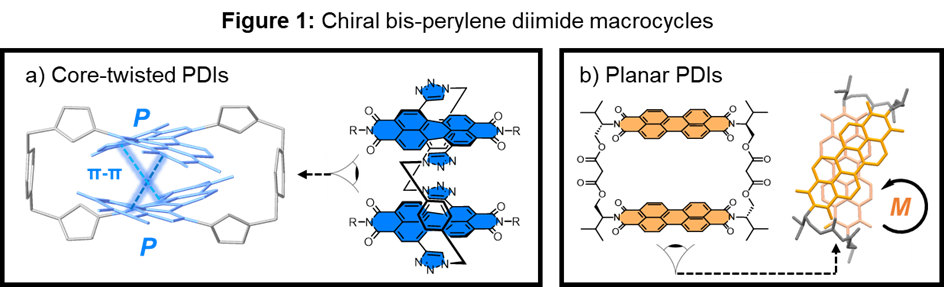
Chiral perylene diimide macrocycles
Perylene diimides (PDIs) are readily functionalisable organic dye molecules and, due to their electron-accepting and photophysical properties, promising building blocks for optoelectronic materials.1 PDIs may also be chiral,2 giving rise to the absorption and/or emission of circularly polarised light. Here, the supramolecular chemistry of PDIs promises the exciting potential to control and amplify these chiroptical properties, which is important for emerging applications in chiroptical sensing, chiral optoelectronics, and spintronics.
Macrocycles provide the opportunity to tune the non-covalent interactions between components preorganised within their cyclic framework.3 As such, we have developed chiral macrocycles to understand how the (π–π) interactions between PDI chromophores impacts their chiral arrangement and in turn their electronic and chiroptical properties (Figure 1). We have explored several sources of chirality in these macrocycles, including those containing helical, core-twisted PDIs4,5 (Figure 1a) and those with point-chiral linkers that give rise to supramolecular chirality (Figure 1b), between planar PDIs.6,7 This presentation will outline how these macrocycles enable us to tune the supramolecular, photophysical and chiroptical properties of PDI assemblies, in both solution and the solid-state.
Reference : 1. C. Huang et al. J. Org. Chem. 2011, 76, 2386 / 2. P. Osswald et al. J. Am. Chem. Soc. 2007, 129, 14319 / 3. P. Spenst et al. J. Photochem. Photobiol. C 2017, 31, 114 / 4. S. E. Penty et al. J. Am. Chem. Soc. 2022, 144, 27, 12290 / 5. S. E. Penty et al. J. Am. Chem. Soc. 2024, 146, 5470 / 6. Hartmann et al. ACIE 2025, e202501122 / 7. Hartmann et al. ACIE 2026, 10.1002/anie.202520567
Doctorants et doctorantes : Valorisez vos travaux !
Inscrivez-vous dès maintenant pour participer à la session posters.
Si vous candidatez également pour une communication orale, merci de déposer votre abstract directement via le formulaire d’inscription. La date limite de soumission des résumés est fixée au 24 Avril 2026.
Modalités d'inscription
L’inscription est obligatoire pour toutes et tous. Pour les doctorants et doctorantes désirant présenter un poster, vous devez simplement l’indiquer lors de votre inscription. Le dépôt d’un abstract est quant à lui réservé exclusivement aux candidats souhaitant effectuer une communication orale.
📜 Inscriptions : jusqu’au Vendredi 24 Avril 2026
⏰ Deadline pour le dépôt des abstracts : jusqu’au Vendredi 24 Avril 2026


